Cell balancing buys extra run time and battery life
Lithium-ion (Li-ion) batteries are the workhorses of the consumer electronics world. Smart phones and laptops would be impractical without them. Now, banks of these cells are enabling electric transportation.
What makes them invaluable is their energy density - two to ten times more than other battery technologies.
But, Lithium-ion's efficiency comes at a price. It's the only mainstream battery chemistry that uses a flammable substance as an electrolyte, so while it's more efficient than battery technologies with water-based electrolytes, such as nickel-cadmium and nickel-metal hydride, it also presents a greater fire risk. If overcharged or overheated, Li-ion cells tend to degrade with disastrous consequences.
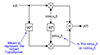
While packaging and quality of the batteries themselves often are the key to preventing such consequences, designing multi-cell battery packs involves additional considerations. The fact is that no two cells, even from the same manufacturing batch, are identical in terms of state of charge (SOC), self-discharge rates, impedance, capacity and temperature characteristics. And that poses a challenge; when cells are placed in series and a battery charger monitors the voltage of the entire string, some cells can be overcharged.
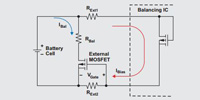
To address this issue, engineers can implement cell balancing, which equalizes the voltages and state-of-charge (SOC) among the cells, when they are at full charge. Balancing can be either voltage-based or SOC-based. It's a non-trivial task that - ideally - aims both to increase the life of a battery pack and to enhance battery safety.
As battery-powered applications proliferate, designing for safe, efficient operation through cell balancing has assumed paramount importance. A classic whitepaper by Texas Instruments examines the tradeoffs between various approaches. It's worth a re-visit.